3551. Ellman's Reagent
Nomenclature
CAS number: 69-78-3
3,3′-Dithiobis[6-nitrobenzoic acid]; 5,5′-dithiobis[2-nitrobenzoic acid]; DTNB. C14H8N2O8S2; mol wt 396.35.
C 42.42%, H 2.03%, N 7.07%, O 32.29%, S 16.18%.
Description and references
Reacts with thiols to produce the yellow dianion of 5-thio-2-nitrobenzoic acid (TNB). Prepn: G. L. Ellman, Arch. Biochem. Biophys. 82, 70 (1959); and spectral properties: P. W. Riddles et al., Anal. Biochem. 94, 75 (1979). Structure determn: E. Shefter, T. I. Kalman, J. Chem. Soc. Chem. Commun. 1969, 1027. Reaction stoichiometry with protein disulfide groups: R. J. Ackerman, J. F. Robyt, Anal. Biochem. 50, 656 (1972). Use in HPLC determn of alkylthiols: K. Kuwata et al., Anal. Chem. 54, 1082 (1982). Molar absorption coefficients for TNB: P. Eyer et al., Anal. Biochem. 312, 224 (2003).
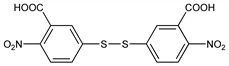
Properties
Yellow crystals from DMF or glacial acetic acid, mp 237-238° (dec) (Ellman); also reported as pale yellow needles from ethanol, mp 239.7-240.3° (dec) (Riddles). d 1.67. Sol in water (pH 8). Absorption max (pH 7.27, 25°): 324 nm (ε 17780).Derivative
Ellman's anion.Nomenclature
CAS number: 77874-90-9
5-Mercapto-2-nitrobenzoic acid ion (2-); 5-thio-2-nitrobenzoate anion; TNB2-. C7H3NO4S; mol wt 197.17.
C 42.64%, H 1.53%, N 7.10%, O 32.46%, S 16.26%.