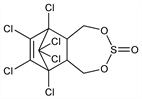
3574. Endosulfan
Nomenclature
CAS number: 115-29-7
6,7,8,9,10,10-Hexachloro-1,5,5a,6,9,9a-hexahydro-6,9-methano-2,4,3-benzodioxathiepin
3-oxide; 1,4,5,6,7,7-hexachloro-5-norbornene-2,3-dimethanol cyclic sulfite; 1,2,3,4,7,7-hexachlorobicyclo[2.2.1]-2-heptene-5,6-bisoxymethylene
sulfite; chlorthiepin; Malix (Bayer CropSci.); Thiodan (Bayer CropSci.); Thionex (Makhteshim-Agan). C9H6Cl6O3S; mol wt 406.93.
C 26.56%, H 1.49%, Cl 52.27%, O 11.80%, S 7.88%.
Description and references
Prepd by reaction of hexachlorocyclopentadiene with cis-butene-1,4-diol to form the bicyclic dialcohol, followed by esterification and cyclization with SOCl2: Geering, Nelson, US 2983732 (1961 to Hooker). Configuration studies: Reimschneider, Wuscherpfenning, Z. Naturforsch. 17b, 585 (1962); Forman et al., J. Org. Chem. 30, 169 (1965). Toxicity study: T. B. Gaines, Toxicol. Appl. Pharmacol. 14, 515 (1969). Review of toxicology and human exposure: Toxicological Profile for Endosulfan (PB2000-108023, 2000) 323 pp.