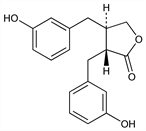
3598. Enterolactone
Nomenclature
Description and references
Phenolic compound having 2,3-dibenzylbutane skeleton. First lignan, q.v., found in animal species. Determn in female urine from humans, vervet monkeys and characterization: S. R. Stitch et al., Nature 287, 738 (1980); K. D. R. Setchell et al., ibid. 740. Structure determn by GC-MS: eidem, Biochem. J. 197, 447 (1981); eidem, Biomed. Mass Spectrom. 10, 227 (1983). 1H-NMR, X-ray crystal structure: G. Cooley et al., J. Chem. Soc. Perkin Trans. 1 1984, 489. Synthesis: M. B. Groen, J. Leemhuis, Tetrahedron Lett. 21, 5043 (1980); G. Cooley et al., ibid. 22, 349 (1981); A. Pelter et al., ibid. 1549; P. A. Ganeshpure, R. Stevenson, Chem. Ind. (London) 1981, 778; A. Pelter et al., J. Chem. Soc. Perkin Trans. 1 1983, 643. Lignan excretion peaks during luteal phase of menstrual cycle: K. D. R. Setchell et al., J. Steroid Biochem. 12, 375 (1980). Excretion as glucuronide conjugate: M. Axelson, K. D. R. Setchell, FEBS Lett. 122, 49 (1980). Biosynthesis by intestinal bacteria: eidem, ibid. 123, 337 (1981). Effect of diet on lignan excretion: H. Aldercreutz et al., Med. Biol. 59, 337 (1981).