3603. Eosine Yellowish—(YS)
Nomenclature
CAS number: 17372-87-1
2′,4′,5′,7′-Tetrabromo-3′,6′-dihydroxyspiro[isobenzofuran-1(3H),9′-[9H]xanthen]-3-one sodium salt (1:2); 2′,4′,5′,7′-tetrabromofluorescein; bromoeosine; tetrabromofluorescein sol; bromofluoresceic acid; eosin; eosine; eosin Y; D & C Red No. 22; C.I. Acid Red 87; C.I. 45380. C20H6Br4Na2O5; mol wt 691.85.
C 34.72%, H 0.87%, Br 46.20%, Na 6.65%, O 11.56%.
Description and references
Xanthene dye capable of photodynamic generation of singlet oxygen. Prepd by bromination of fluorescein: Colour Index vol. 4 (3rd ed., 1971) p 4426. Photodynamic injury to leaf tissue and inhibition of photosynthesis: J. P. Knox, A. D. Dodge, Planta 164, 22; 30 (1985). Review: H. J. Conn's Biological Stains, R. W. Horobin, J. A. Kiernan, Eds. (BIOSIS Scientific, Oxford, 10th ed., 2002), pp 231-233.
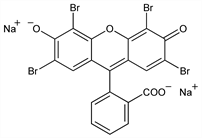
Properties
Red crystals with bluish tinge, or brownish-red powder. Freely sol in water, less in alcohol. Insol in ether. The concd aq soln is deep brownish-red, the dilute (1:500) soln is yellowish-red with greenish fluorescence; the alcoholic soln exhibits a strong green fluorescence.Derivative
Free acid.Nomenclature
CAS number: 15086-94-9
Eosine acid; D & C Red No. 21; C.I. Solvent Red 43; C.I. 45380:2. C20H8Br4O5; mol wt 647.89.
C 37.08%, H 1.24%, Br 49.33%, O 12.35%.