3643. Equilin
Nomenclature
Description and references
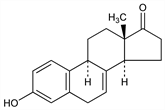
Steroidal hormone isolated from urine of pregnant mares: Girard et al., Compt. Rend. 195, 981 (1932); Cartland, Meyer, J. Biol. Chem. 112, 9 (1935/6). Structure: Serini, Logemann, Ber. 71, 186 (1938); Pearlman, Wintersteiner, J. Biol. Chem. 130, 35 (1939). Separation from estrone: Serini, Logemann, US 2221340 (1941 to Schering). Synthesis: Zderic et al., J. Am. Chem. Soc. 80, 2596 (1958); Bagli et al., Tetrahedron Lett. 1964, 387; Stein et al., ibid. 1966, 5015; eidem, Tetrahedron 26, 1917 (1970). Prepn from testosterone by bacterial fermentation: Bowers et al., US 3067212 (1962 to Syntex). Prepn by conversion of equilenin: Bailey et al., Chem. Commun. 1967, 1253; Marshall, Deghenghi, Can. J. Chem. 47, 3127 (1969). Component of Conjugated Estrogenic Hormones, q.v. Review of synthetic studies: Taub, “Naturally Occurring Aromatic Steroids” in The Total Synthesis of Natural Products vol. 2, J. ApSimon, Ed. (John Wiley & Sons, New York, 1973) pp 664-670.
Properties
Orthorhombic sphenoidal plates from ethyl acetate, mp 238-240°. [α]D25 +308° (c = 2 in dioxane); [α]D25 +325° (c = 2 in alc). uv max: 283-285 nm. Soluble in alcohol, dioxane, acetone, ethyl acetate, in other organic solvents; sparingly sol in water.Derivative
Benzoate.Properties
mp 196-197°.Derivative
Methyl ether.Properties
Needles from alc, mp 161-162°.Therapeutic Category
Estrogen.
Keywords
Estrogen; Steroidal