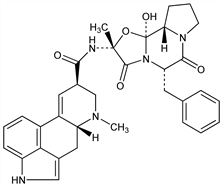
3661. Ergotamine
Nomenclature
Description and references
Vasoconstrictor found in ergot of Central Europe. Extraction procedure: Stoll, Helv. Chim. Acta 28, 1283 (1945). Pharmacology: E. Rothlin, Schweiz. Med. Wochenschr. 76, 1254 (1946). Structure: Stoll et al., ibid. 34, 1544 (1951). Total synthesis: Hofmann et al., Experientia 17, 206 (1961). Stereochemistry: Hofmann et al., ibid. 46, 2306 (1963). Comprehensive description: B. Kreilgard, Anal. Profiles Drug Subs. 6, 113-159 (1977). LC determn in tablets: U. R. Cieri, J. Assoc. Off. Anal. Chem. 70, 538 (1987); GC/MS determn in plasma: N. Feng et al., J. Chromatogr. 575, 289 (1992). Bioavailablity and efficacy in migraine: V. Ala-Hurula, Headache 22, 167 (1982). Review of clinical pharmacokinetics and treatment of headache: V. L. Perrin, Clin. Pharmacokinet. 10, 334-352 (1985). Review of teratogenic risk: G. V. Raymond, Teratology 51, 344-347 (1995).
Properties
Elongated prisms from benzene. Very hygroscopic. Darkens and dec on exposure to air, heat and light. Dec 212-214°. [α]D20 -160° (chloroform). Sol in about 70 parts methanol, 150 parts acetone, 300 parts alcohol; freely sol in chloroform, pyridine, glacial acetic acid; moderately sol in ethyl acetate; slightly in benzene. Almost insol in water, petr ether. LD50 in mice, rats, rabbits (mg/kg): 62, 80, 3 i.v.; in cats: 11 s.c. (Rothlin).Derivative
Hydrochloride.Properties
Rectangular plates from 90% alc, mp 212° (dec). Sol in water-alcohol mixtures; sparingly in water or alcohol.Derivative
Tartrate.Nomenclature
Properties
Solvated crystals, e.g. the dimethanolate, heavy rhombic plates from methanol, mp 203° (dec). [α]D25 -125 to -155° (c = 0.4 in chloroform). One gram dissolves in 500 ml water or 500 ml alc. Protect from heat and light.Therapeutic Category
Antimigraine.
Therapeutic Category (Veterinary)
Tartrate has been used as an oxytocic.Keywords
Antimigraine; Serotonin Receptor Agonist