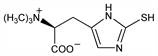3663. Ergothioneine
Nomenclature
CAS number: 497-30-3
(αS)-α-Carboxy-2,3-dihydro-N,N,N-trimethyl-2-thioxo-1H-imidazole-4-ethanaminium inner salt; [1-carboxy-2-[2-mercaptoimidazol-4-yl]ethyl]trimethylammonium
hydroxide inner salt; l(+)-ergothioneine; thioneine; thiolhistidine-betaine; thiasine; sympectothion. C9H15N3O2S; mol wt 229.30.
C 47.14%, H 6.59%, N 18.33%, O 13.95%, S 13.98%.
Description and references
First discovered in the sclerotia of the ergot fungus, Claviceps purpurea, by Tanret, J. Pharm. Chim. 30, 145 (1909). Has since been found to occur in blood, semen and various mammalian tissues, principally liver and kidneys. Biosynthesis from histidine by Claviceps purpurea cultures: Heath, Wildy, Nature 179, 196 (1957); also produced by Neurospora crassa: Melville et al., Fed. Proc. 15, 314 (1956). Chemical synthesis: Heath et al., J. Chem. Soc. 1951, 2215. Occurrence in the Linulus polyphemus L. (king crab): Ackermann, List, Z. Physiol. Chem. 313, 30 (1958). Review: Melville, Vitam. Horm. 17, 155 (1959).