3668. Eriodictyol
Nomenclature
CAS number: 552-58-9
2-(3,4-Dihydroxyphenyl)-2,3-dihydro-5,7-dihydroxy-4H-1-benzopyran-4-one; 3′,4′,5,7-tetrahydroxyflavanone. C15H12O6; mol wt 288.25.
C 62.50%, H 4.20%, O 33.30%.
Description and references
Isoln from Eriodictyon californicum (H. & A.) Greene, Hydrophyllaceae: Geissman, J. Am. Chem. Soc. 62, 3258 (1940); from lemon: Mager, Z. Physiol. Chem. 274, 109 (1942); Horowitz, J. Am. Chem. Soc. 79, 6561 (1957); US 2857318 (1958 to U.S. Dept. Agr.). Structure and synthesis: Reichel et al., Ann. 550, 146 (1942); Zemplén et al., Ber. 76B, 1112 (1943); Pew, J. Org. Chem. 27, 2935 (1962). Synthesis: G. Wurm, U. Geres, Arch. Pharm. 315, 183 (1982). See also Bioflavanoids.
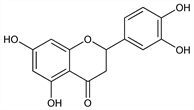
Properties
Needles with 1 1/2 H2O from dil alc, dec 257° (rapid heating). After drying in vacuo at 100° for 6 hours, dec 267°. uv max (alc): 290, 326 nm (log ε 2.54, 2.16). Sparingly sol in boiling water, hot alcohol, ether, glacial acetic acid; sol in dil alkalies.Derivative
7-l-Rhamnoside.Nomenclature
Eriodictin.C21H22O10; mol wt 434.39.
C 58.06%, H 5.10%, O 36.83%.
Description and references
Isoln from citrin: Bruckner, Szent-Gy"orgyi, Nature 138, 1057 (1936). Structure: Mager, loc. cit.