3673. Ertapenem
Nomenclature
Description and references
Group 1 carbapenem antibiotic. Prepn: M. J. Betts et al., WO 9315078; eidem, US 5478820 (1993, 1995 both to Zeneca). Large-scale synthesis: J. M. Williams et al., J. Org. Chem. 70, 7479 (2005). Pharmacokinetics in primates: J. G. Sundelof et al., Antimicrob. Agents Chemother. 41, 1743 (1997). In vivo efficacy and pharmacokinetics: C. J. Gill et al., ibid. 42, 1996 (1998). In vitro efficacy and β-lactamase stability: J. Kohler et al., ibid. 43, 1170 (1999). Stability in aq solns: M. Zajac et al., J. Pharm. Biomed. Anal. 43, 445 (2007). HPLC determn in plasma and urine: D. G. Musson et al., J. Chromatogr. B 720, 99 (1998); of residual crystallization solvents: T. K. Natishan, Y. Wu, J. Chromatogr. A 800, 275 (1998). Series of articles on pharmacology and clinical experience in complicated community-acquired infections: J. Antimicrob. Chemother. 53, Suppl. S2, ii1-ii86 (2004). Review of clinical experience: O. Burkhardt et al., Expert Opin. Pharmacother. 8, 237-256 (2007).
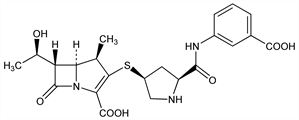
Derivative
Sodium salt.Nomenclature
Properties
White to off-white hygroscopic, weakly crystalline powder. Sol in water, 0.9% sodium chloride soln. Practically insol in ethanol; insol in isopropyl acetate, THF.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Carbapenems