3695. Esaprazole
Nomenclature
Description and references
Prepn: C. Corvi-Mora, DE 2702537; idem, US 4123530 (1977, 1978 both to Camillo Corvi). Metabolism in animals and man: P. Ventura et al., Farmaco Ed. Prat. 39, 190 (1984). Cytoprotective activity: B. Lumachi, R. Scuri, Drugs Exp. Clin. Res. 11, 693 (1985); and pharmacology: R. Scuri et al., Boll. Chim. Farm. 123, 425 (1983). Pharmacology: G. Coruzzi et al., Farmaco Ed. Prat. 40, 313 (1985); G. Coruzzi et al., ibid. 324. Antisecretory activity in humans: L. Erembourg et al., Int. J. Clin. Pharmacol. Res. 7, 95 (1987). HPLC determn in biological fluids: M. A. Girometta et al., J. Chromatogr. 383, 85 (1986). Clinical trial in comparison with cimetidine: L. Capurso et al., Clin. Trials J. 23, 293 (1986).
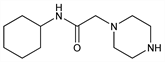
Properties
Residue, mp 111-112°. bp0.5 190°. LD50 in mice (mg/kg): 1974 orally; 271 i.v.; in rats (mg/kg): 3900 orally (Corvi-Mora).Derivative
Hydrochloride.Nomenclature
CO-1063.Therapeutic Category
Antiulcerative.
Keywords
Antiulcerative