3716. Ethacridine
Nomenclature
Description and references
Prepn: DE 364033; DE 364037; DE 393411; DE 395683 (1922); Frdl. 14, 804, 807-812; A. Albert, W. Gledhill, J. Soc. Chem. Ind. 61, 159 (1942). Toxicity: S. D. Rubbo, Br. J. Exp. Pathol. 28, 1 (1947); N. J. Joshi et al., Indian J. Exp. Biol. 16, 1038 (1978). Pharmacokinetics: T. J. Rising et al., Arzneim.-Forsch. 27, 872 (1977). Use in protein analysis: R. H. Yue et al., Thromb. Diath. Haemorrh. 31, 439 (1974); F. Franek, Methods Enzymol. 121, 631-638 (1986); in serological detection of Brucella sp.: J. D. Huber, P. Nicoletti, Am. J. Vet. Res. 47, 1529 (1986). Clinical evaluation in diarrhea: N. Madanagopalan et al., Curr. Ther. Res. 18, 546 (1975); R. Raedsch et al., Klin. Wochenschr. 69, 863 (1991); as an abortifacient: S. Gardo, M. Nagy, Arch. Gynecol. Obstet. 247, 39 (1990).
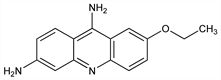
Properties
Orange-yellow crystals from 50% alc. mp 226°.Derivative
Lactate monohydrate.Nomenclature
Properties
Light yellow crystals from 90% alcohol + ether. Darkens at 200°, mp 235°. Slowly sol in 15 parts water; sol in 9 parts boiling water; in 110 parts alcohol at 22°, in 100 parts boiling alcohol. Solns are yellow, fluorescent and stable to boiling. LD50 s.c. in mice: 0.12 g/kg (Rubbo).Use
Reagent in serological testing and protein chemistry.Therapeutic Category
Antiseptic. Abortifacient.
Keywords
Antiseptic/Disinfectant; Dyes