3806. Ethyl Ether
Nomenclature
CAS number: 60-29-7
1,1′-Oxybisethane; ethoxyethane; ether; diethyl ether; ethyl oxide; diethyl oxide; sulfuric ether; anesthetic ether. C4H10O; mol wt 74.12.
C 64.82%, H 13.60%, O 21.59%.
Description and references
Produced on a large scale by dehydration of ethanol or by hydration of ethylene, both processes being carried out in the presence of sulfuric acid. Review of mfg processes: Himmler in Ullmanns Encyklop"adie der technischen Chemie vol. 5 (1954) pp 777-782; D. E. Keeley in Kirk-Othmer Encyclopedia of Chemical Technology vol. 9 (Wiley-Interscience, New York, 3rd ed., 1980) pp 381-393.
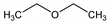
Properties
Mobile, very volatile, highly flammable liq. Explosive. Vapor heavier than air. Characteristic, sweetish, pungent odor, more agreeable than chloroform. Burning taste. Tends to form explosive peroxides under the influence of air and light, esp when evaporation to dryness is attempted. Peroxides may be removed from ether by shaking with 5% aq ferrous sulfate soln. Addition of naphthols, polyphenols, aromatic amines, and aminophenols has been proposed for the stabilization of ethyl ether. d40 0.7364; d410 0.7249; d420 0.7134; d430 0.7019. Vapor density 2.55 (air = 1.0). mp -116.3° (stable crystals); mp -123.3° (metastable crystals). bp760 34.6°; bp400 17.9°; bp200 2.2°; bp100 -11.5°; bp10 -48.1°; bp1.0 -74.3°. Satd vapor press. at 0°: 184.9 mm; at 10°: 290.8 mm; at 20°: 439.8 mm; at 50°: 1276 mm; at 70°: 2304 mm. Critical temp 192.7°; crit press. 35.6 atm. Flash pt, closed cup: -49°F (-45°C). Air-ether mixtures containing more than 1.85 volume-% of ether vapor, are explosive hazards. Autoignition temp 180-190°. nD15 1.35555. Dielectric constant at 26.9° and 85.8 kilocycles = 4.197; good insulator. When shaken under absolutely dry conditions ether can generate enough static electricity to start a fire. Surface tension at 20°: 17.06 dynes/cm. Viscosity at 20°: 0.2448 cP. Heat of vaporization at 30°: 89.80 cal/g. Produces considerable coldness on quick evaporation. Heat of formation -907 cal/g; heat of combustion -8.807 kcal/g. Ether is slightly sol in water and water is slightly sol in ether. A satd water soln contains 8.43% (w/w) of ether at 15° and 6.05% (w/w) at 25°. Ether satd with water contains 1.2% H2O at 20°. Soly in water increased by HCl. Sol in concd hydrochloric acid. May explode when brought in contact with anhydr nitric acid. Miscible with lower aliphatic alcohols, benzene, chloroform, petr ether, other fat solvents, many oils. Azeotrope with water (1.3%), bp 34.2°.Caution
Potential symptoms of overexposure are dizziness; drowsiness; headache, excitedness and narcosis; nausea, vomiting; irritation of eyes, upper respiratory system and skin. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 140.Use
Solvent for waxes, fats, oils, perfumes, alkaloids, gums. Excellent solvent for nitrocellulose when mixed with alc. Important reagent in organic syntheses, esp in Grignard and Wurtz type reactions. Easily removable extractant of active principles (hormones, etc.) from plant and animal tissues. In the manuf of gun powder. As primer for gasoline engines.Therapeutic Category
Anesthetic (inhalation).
Therapeutic Category (Veterinary)
Anesthetic (inhalation). Has been used orally in colic, subcutaneously as a stimulant.Keywords
Anesthetic (Inhalation)