3886. Etoposide
Nomenclature
Description and references
DNA topoisomerase II inhibitor. Semi-synthetic deriv of podophyllotoxin, related structurally to teniposide, q.q.v. Prepn: C. Keller-Juslén et al., US 3524844 (1970 to Sandoz); idem et al., J. Med. Chem. 14, 936 (1971). Teratogenicity and cytogenicity study: S. M. Sieber et al., Teratology 18, 31 (1978). Review of pharmacokinetics and assay methods: P. I. Clark, M. L. Slevin, Clin. Pharmacokinet. 12, 223-252 (1987). Comprehensive description: J. J. M. Holthuis et al., Anal. Profiles Drug Subs. 18, 121-151 (1989). Review of pharmacology and clinical experience: J. D. Hainsworth, F. A. Greco, Ann. Oncol. 6, 325-341 (1995); S. Joel, Cancer Treat. Rev. 22, 179-221 (1996).
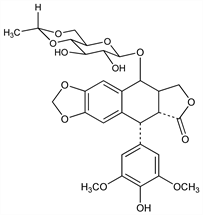
Properties
Crystals from methanol, mp 236-251°. [α]D20 -110.5° (c = 0.6 in chloroform). uv max (abs methanol): 283 nm (ε 4245). pKa 9.8. Very sol in methanol, chloroform; slightly sol in ethanol; sparingly sol in water.Derivative
Phosphate.Nomenclature
Description and references
Review: A. H. I. Witterland et al., Pharm. World Sci. 18, 163-170 (1996).
Properties
Sol in water. Practically insol in organic solvents.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Alkaloids/Natural Products; Podophyllum Derivatives; Topoisomerase II Inhibitor