3898. Eugenol
Nomenclature
CAS number: 97-53-0
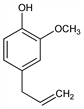
2-Methoxy-4-(2-propenyl)phenol; 4-allyl-2-methoxyphenol; allylguaiacol; eugenic acid; caryophyllic acid. C10H12O2; mol wt 164.20.
C 73.15%, H 7.37%, O 19.49%.
Description and references
Obtained from many natural sources: Beilstein vol. 6, 961. Prepn: Claisen, Ann. 418, 113 (1919); from oil of cloves: Waterman, Priester, Rec. Trav. Chim. 48, 1272 (1929). Toxicity study: E. C. Hagan et al., Toxicol. Appl. Pharmacol. 7, 18 (1965).
Properties
Colorless or pale yellow liquid, bp 255°. Darkens and thickens on exposure to air. Odor of cloves; spicy, pungent taste. mp -9.2 to -9.1°. d420 1.0664. nD20 1.5410. Practically insol in water. Misc with alcohol, chloroform, ether, oils. One ml dissolves in 2 ml 70% alcohol; sol in glacial acetic acid, in aq fixed alkali hydroxide solns. Ferric chloride, potassium permanganate. LD50 in rats, mice (mg/kg): 2680, 3000 orally (Hagan).Derivative
Benzoate.Nomenclature
O-Benzoyleugenol.C17H16O3; mol wt 268.31.
C 76.10%, H 6.01%, O 17.89%.
Properties
Crystals, mp 69-70°. bp 360°. Freely sol in benzene, chloroform, ether, hot alcohol. Practically insol in water.Use
In perfumery instead of oil of cloves; manuf vanillin. As insect attractant.Therapeutic Category
Analgesic (dental).
Keywords
Analgesic (Dental)