3900. Euparin
Nomenclature
CAS number: 532-48-9
1-[6-Hydroxy-2-(1-methylethenyl)-5-benzofuranyl]ethanone; 6-hydroxy-2-isopropenyl-5-benzofuranyl methyl ketone; 5-acetyl-6-hydroxy-2-isopropenylbenzofuran. C13H12O3; mol wt 216.23.
C 72.21%, H 5.59%, O 22.20%.
Description and references
From roots of Eupatorium purpureum L., E. cannabinum L., Compositae: Kamthong, Robertson, J. Chem. Soc. 1939, 925; Jermanowska, C.A. 48, 5848h (1954). Structure: Kamthong, Robertson, J. Chem. Soc. 1939, 933. Synthesis: Ramachandran et al., J. Org. Chem. 28, 2744 (1963). Review: Gizycki, Pharmazie 6, 686 (1951).
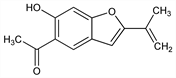
Properties
Yellow needles from alcohol, mp 121-122°. uv max (ethanol): 263, 358 nm (ε 34400, 5900). Sol in alcohol, benzene, chloroform, ether. Practically insol in water, alkali.Derivative
Acetate.C15H14O4; mol wt 258.27.
C 69.76%, H 5.46%, O 24.78%.
Properties
Prisms from petr ether, mp 80°.Derivative
O-Methyl deriv.Nomenclature
O-Methyleuparin; 2-isopropenyl-6-methoxy-5-benzofuranyl methyl ketone.C14H14O3; mol wt 230.26.
C 73.03%, H 6.13%, O 20.85%.