4094. Flavopiridol
Nomenclature
CAS number: 146426-40-6
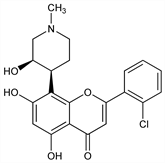
rel-()-2-(2-Chlorophenyl)-5,7-dihydroxy-8-[(3R,4S)-3-hydroxy-1-methyl-4-piperidinyl]-4H-1-benzopyran-4-one; cis-()-2-(2-chlorophenyl)-5,7-dihydroxy-8-[4′-(3′-hydroxy-1′-methyl)-piperidinyl]-4H-1-benzopyran-4-one. C21H20ClNO5; mol wt 401.84.
C 62.77%, H 5.02%, Cl 8.82%, N 3.49%, O 19.91%.
Description and references
Flavone inhibitor of cyclin-dependent kinases; the ()-cis form induces apoptosis in certain tumor cells. Prepn: S. L. Kattige et al., EP 241003; eidem, US 4900727 (1987, 1990 both to Hoechst). Properties and formulation study: R.-M. Dannenfelser et al., PDA J. Pharm. Sci. Technol. 50, 356 (1996). Mode of action study: S. Brüsselbach et al., Int. J. Cancer 77, 146 (1998). Clinical evaluation in refractory cancers: A. M. Senderowicz et al., J. Clin. Oncol. 16, 2986 (1998). Clinical pharmacokinetics: J. P. Thomas et al., Cancer Chemother. Pharmacol. 50, 465 (2002). Review of chemistry, pharmacology, and antitumor spectrum: H. H. Sedlacek et al., Int. J. Oncol. 9, 1143-1168 (1996).
Derivative
Hydrochloride.Nomenclature
CAS number: 131740-09-5
L-86-8275; HMR-1275; NSC-649890. C21H20ClNO5.HCl; mol wt 438.30.
C 57.55%, H 4.83%, Cl 16.18%, N 3.20%, O 18.25%.
Properties
mp 169.5± 0.5° (Dannenfelser). Also reported as yellow powder, mp 188° containing 4.73-6.2% water of crystallization (Sedlacek). pKa 5.68 ± 0.06. [α]24D 1.73 to 3.9°. Soly (mg/ml): water, D5W, and normal saline all <5 at pH 4. Soly (mg/ml): ethanol 10.1; PEG 400 >73.8; propylene glycol >88.1; M-pyrol very much greater than 103.0; τ-butyrolactone very much greater than 105.5.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic