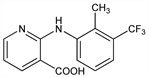
4147. Flunixin
Nomenclature
Description and references
Cyclooxygenase inhibitor. Prepn: M. H. Sherlock, N. Sperber, BE 679271; eidem, US 3337570 (1966, 1967 both to Schering). Prepn of the acid and meglumine salt: M. H. Sherlock, BE 812772; idem, US 3839344 (both 1974 to Schering). Pharmacodynamics in horses: M. M. Hardee, J. N. Moore, Res. Vet. Sci. 40, 152 (1986); and pharmacokinetics: L. R. Soma et al., J. Vet. Pharmacol. Ther. 15, 292 (1992). GC determn in urine: M. Johansson, E. -L. Anlér, J. Chromatogr. 427, 55 (1988). Clinical evaluation in horses: J. W. Houdeshell, P. W. Hennessey, J. Equine Med. Surg. 1, 57 (1977). Experimental use in food-producing animals: M. Kopcha, A. S. Ahl, J. Am. Vet. Med. Assoc. 194, 45 (1989). Comparative toxicological study in horses: C. G. MacAllister et al., ibid. 202, 71 (1993).