4159. Fluorescein
Nomenclature
Description and references
Prepd by heating phthalic anhydride with resorcinol: Fischer, Bollmann, J. Prakt. Chem. 104, 123 (1922); McKenna, Sowa, J. Am. Chem. Soc. 60, 124 (1938). Structure: Ramart-Lucas, Compt. Rend. 205, 864 (1937); Nagase et al., J. Pharm. Soc. Jpn. 73, 1033, 1039 (1953). Review of synthesis, properties and histological use: R. F. Steiner, H. Edelhoch, Chem. Rev. 62, 457 (1962). Use as label in immunoassays: E. F. Ullman et al., J. Biol. Chem. 251, 4172 (1976); Y. Suzuki et al., Jpn. J. Exp. Med. 49, 179 (1979). Toxicity studies in fish: L. L. Marking, Prog. Fish Cult. 31, 139 (1969). Toxicity data: S. L. Yankell, J. J. Loux, J. Periodontol. 48, 228 (1977). See also: Colour Index vol. 4 (3rd ed., 1971) p 4424; H. J. Conn's Biological Stains, R. D. Lillie, Ed. (Williams & Wilkins, Baltimore, 9th ed., 1977) p 337.
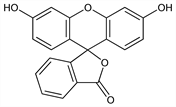
Properties
Yellowish-red to red powder. mp 314-316° in sealed tube, with decompn. Insol in water, benzene, chloroform, ether. Sol in hot alcohol or glacial acetic acid; also sol in alkali hydroxides or carbonates with a bright green fluorescence appearing red by transmitted light. Absorption max: 493.5, 460 nm.Derivative
Disodium salt.Nomenclature
Properties
Hygroscopic orange-red powder. Freely sol in water with yellowish-red color and intense yellowish-green fluorescence perceptible down to a dil of 0.02 ppm under uv light. The fluorescence disappears when the soln is made acid, and reappears when the soln is again made neutral or alkaline. Absorption max (water): 493.5 nm. Slightly sol in alc. LD50 in mice, rats (mg/kg): 4738, 6721 orally (Yankel, Loux).Use
In examining subterranean waters. Serves to ascertain source of springs, connections between streams and sea, determining approx vol of water delivered by a spring, detecting source of contamination of drinking water, infiltration of soil with waste waters of factories. In externally applied drugs and cosmetics. Analytical reagent (protein label). Clinical reagent (immunohistological stain, immunofluorescent label).Therapeutic Category
Diagnostic aid (corneal trauma indicator), ophthalmic angiography, contact lens fitting.
Therapeutic Category (Veterinary)
Diagnostic aid (corneal lesions, intraocular inflammation).Keywords
Diagnostic Aid