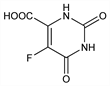
4176. 5-Fluoroorotic Acid
Nomenclature
Description and references
Pyrimidine precursor; selectively toxic to yeast cells that synthesize the enzyme orotidine-5′-P decarboxylase. Prepn: R. Duschinsky et al., J. Am. Chem. Soc. 79, 4559 (1957); GB 806584 (1958 to Hoffmann-La Roche); R. Duschinsky, C. Heidelberger, US 2948725 (1960). Improved synthesis: D. H. R. Barton et al., J. Chem. Soc. Perkin Trans. 1 1974, 2095; and mass spectrum: S. N. Alam et al., Acta Pharm. Suec. 12, 375 (1975). Antitumor activity: C. Heidelberger et al., Nature 179, 663 (1957). Inhibition of RNA synthesis: C. T. Garrett et al., Arch. Biochem. Biophys. 155, 342 (1973). Clinical evaluation of antimycotic activity: K. Nikolova et al., Methods Find. Exp. Clin. Pharmacol. 9, 85 (1987). Review of in vitro use in the positive selection of genetically transformed yeast cells: J. D. Boeke et al., Methods Enzymol. 154, 164-175 (1987).