4181. Fluorouracil
Nomenclature
Description and references
Pyrimidine analog; nucleotide metabolites inhibit DNA synthesis and incorporate into RNA. Prepn: R. Duschinsky et al., J. Am. Chem. Soc. 79, 4559 (1957); C. Heidelberger, R. Duschinsky, US 2802005 (1957); D. H. R. Barton et al., J. Org. Chem. 37, 329 (1972); O. Miyashita et al., Chem. Pharm. Bull. 29, 3181 (1981). Site of action: F. Maley, G. F. Maley, FEBS Symp. 57, 21 (1979). Clinical trial in actinic keratoses: C. S. Jury et al., Br. J. Dermatol. 153, 808 (2005); as an adjuvant in rectal cancer: S. R. Smalley et al., J. Clin. Oncol. 24, 3542 (2006). Comprehensive description: B. C. Rudy, B. Z. Senkowski, Anal. Profiles Drug Subs. 2, 221-244 (1973); S. M. Bayomi, A. A. Al-Badr, ibid. 18, 599-639 (1989). Review of pharmacokinetics: W. Sadee, C. G. Wong, Clin. Pharmacokinet. 2, 437-450 (1977); of clinical trials: R. M. Hansen, Cancer Invest. 9, 637-642 (1991); in colon cancer: W. Kelder et al., Expert Rev. Anticancer Ther. 6, 785-794 (2006).
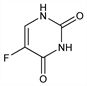
Properties
Crystals from water or methanol-ether, mp 282-283° (dec). Sublimes (0.1 mm) 190-200°. uv max (0.1N HCl): 265-266 nm (ε 7070). Sparingly sol in water; slightly sol in alcohol.Therapeutic Category
Antineoplastic.
Therapeutic Category (Veterinary)
Antineoplastic.Keywords
Antineoplastic; Antimetabolites; Pyrimidine Analogs