4222. Folinic Acid
Nomenclature
Description and references
Intermediate product of the metabolism of folic acid; the active form into which that acid is converted in the body, ascorbic acid being a necessary factor in the conversion process. First reported as the Leuconostoc citrovorum 8081 growth factor: H. E. Sauberlich, C. A. Baumann, J. Biol. Chem. 176, 165 (1948). Isoln from houseflies: S. Miller, A. S. Perry, Life Sci. 4, 1573 (1965). Prepn: J. A. Brockman, Jr., et al., J. Am. Chem. Soc. 72, 4325 (1950); E. Khalifa et al., Helv. Chim. Acta 63, 2554 (1980). Isomers: D. B. Cosulich et al., J. Am. Chem. Soc. 74, 4215 (1952). Structure: May et al., ibid. 73, 3067 (1951); Pohland et al., ibid. 3247. Manuf: Shive, US 2741608 (1956 to Res. Corp.). Stereoselective synthesis: J. Owens, et al., J. Chem. Soc. Perkin Trans. 1 7, 871 (1993). Used as an antidote to folic acid antagonists such as methotrexate, q.v., which block the conversion of folic acid into folinic acid. Review of clinical combination therapy with methotrexate: J. R. Bertino et al., Ann. N.Y. Acad. Sci. 186, 486-495 (1971). Pharmacokinetics: P. F. Nixon, Clin. Exp. Pharmacol. Physiol. Suppl. 5, 35 (1979). Comprehensive description: L. O. Pont et al., Anal. Profiles Drug Subs. 8, 315-350 (1979). Review of clinical synergy with fluorouracil in cancer: R. J. DeLap, Yale J. Biol. Med. 61, 23-34 (1988).
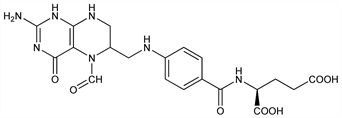
Properties
Crystals, dec 240-250°. uv max (0.1N NaOH): 282 nm (% T = 27.0 for 10 mg/l). [α]D20 +14.26° (c = 3.42 as anhydr Ca salt). pKa (3 groups): 3.1, 4.8, and 10.4. Sparingly sol in water. pH of satd aq soln 2.8-3.0 at which pH partial decompn takes place. More stable at neutral or mildly alkaline pH.Derivative
Calcium salt pentahydrate.Nomenclature
Properties
Off-white to light beige amorphous, odorless powder, freely sol in water. Practically insol in alc. [α]D21 +14.9° (c = 1 in water).Derivative
l-Form calcium salt.Nomenclature
Properties
[α]D20 15.1° (c = 1.82).Therapeutic Category
Antidote to folic acid antagonists; antianemic (folate deficiency).
Keywords
Antidote (Folic Acid Antagonists); Antineoplastic Adjunct; Folic Acid Replenisher