4225. Fomecins
Description and references
Antibacterial substances produced by several strains of the basidiomycete Fomes juniperinus Schrenk. Isoln of fomecin A (major) and fomecin B (minor) from cultures grown in corn steep liquor: M. Anchel et al., Proc. Natl. Acad. Sci. USA 38, 655 (1952). Structures: T. C. McMorris, M. Anchel, Can. J. Chem. 42, 1595 (1964). Synthesis of B: S. M. Al-Mousawi et al., Bull. Soc. Chim. Belg. 88, 883 (1979); of A and B: K. Hayashi et al., Chem. Pharm. Bull. 28, 1971 (1980).
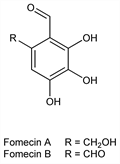
Derivative
Fomecin A.Nomenclature
2,3,4-Trihydroxy-6-(hydroxymethyl)benzaldehyde.C8H8O5; mol wt 184.15.
C 52.18%, H 4.38%, O 43.44%.
Properties
Cream-colored to orange crystals from ethanol-water, ethanol-benzene, acetone-benzene, or ethyl acetate. Dec above 160° without melting. Optically inactive in ethanol. uv max (ethanol): 241, 304 nm (ε 10800, 15300). Weakly acidic. Sparingly sol in water (1 mg/ml); slightly more sol in ethanol, acetone, ethyl acetate. Less sol in chloroform, benzene. Aq solns at neutral or acid pH are stable. The activity is lost around pH 8.Derivative
Fomecin B.Nomenclature
3,4,5-Trihydroxy-1,2-benzenedicarboxaldehyde.C8H6O5; mol wt 182.13.
C 52.76%, H 3.32%, O 43.92%.