4244. Formononetin
Nomenclature
Description and references
Isoln from soy-bean meal (Soja hispida): Walz, Ann. 489, 118 (1931); from clover species Trifolium subterraneum L. and T. pratense L., Leguminosae in which it is the major estrogenic factor: Bradbury, White, J. Chem. Soc. 1951, 3447; Bate-Smith, Swain, Chem. Ind. (London) 1953, 1127. Identity with biochanin B: Bose, J. Sci. Ind. Res. 15B, 325 (1956). Structure: Baker et al., J. Chem. Soc. 1933, 274. Synthesis: Wessely et al., Ber. 66, 685 (1933); Kagal et al., Tetrahedron Lett. 1962, 593. Biological half-life in Cicer arietinum L., Papilionatae: N. Amrhein, E. Diederich, Naturwissenschaften 67, 40 (1980). HPLC analysis: R. E. Carlson, J. Chromatogr. 198, 193 (1980). 13C-NMR study: H. C. Jha et al., Can. J. Chem. 58, 1211 (1980). Mutagenicity study: R. M. Bartholomew et al., Mutat. Res. 78, 317 (1980).
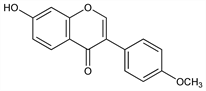
Properties
Needles from alcohol, mp 258°. uv max (ethanol): 250, 300 nm (ε 27440, 11240).Derivative
7-Glucoside.Nomenclature
Ononin; 4′-methyldaidzin.Description and references
From Ononis spinosa L., Leguminosae: Hlasiwetz, J. Prakt. Chem. 65, 415 (1855). Synthesis: Farkas, Varady, Ber. 92, 819 (1959).