4249. Fortimicins
Description and references
Aminoglycoside antibiotic complex produced by Micromonospora olivoasterospora. The main components, fortimicins A and B, both exhibit broad-spectrum antibacterial activity. Isoln of A: T. Nara et al., JP Kokai 75 29789 (to Kyowa); eidem, US 3976768 (1976 to Abbott). Isoln of B: eidem, DE 2418349 (1974 to Kyowa); eidem, US 3931400 (1976 to Abbott). Isoln and biological properties of A and B: eidem, J. Antibiot. 30, 533 (1977); physical and chromatographic properties: R. Okachi et al., ibid. 541. Structures of A and B: R. S. Egan et al., ibid. 552. In vitro activity: R. Girolami, J. S. Stamm, ibid. 564. Isoln and properties of minor components, fortimicins C, D, KE: M. Sugimoto et al., ibid. 32, 868 (1979). Synthesis of B: T. Suami, Y. Honda, Chem. Lett. 1980, 641; Y. Honda, T. Suami, Bull. Chem. Soc. Jpn. 55, 1156 (1982).
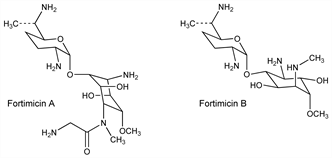
Derivative
Fortimicin A.Nomenclature
Properties
White amorphous powder, mp >200° (dec). [α]D25 +87.5° (c = 0.1 in water). Sol in water and lower alcohols. Insol in organic solvents. LD50 (of the sulfate salt) in mice (mg/kg): 380 i.v.; 400 s.c. (Nara, 1977).Derivative
Fortimicin B.Nomenclature
Properties
White amorphous powder, mp 101-103°. [α]D25 +22.2° (c = 0.1 in water). Sol in water and lower alcohols. Insol in organic solvents.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); Aminoglycosides