4253. Fosfestrol
Nomenclature
CAS number: 522-40-7
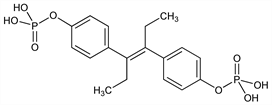
4,4′-[(1E)-1,2-Diethyl-1,2-ethenediyl]bisphenol
bis(dihydrogen phosphate); α,α′-diethyl-4,4′-stilbenediol diphosphoric acid ester; diethylstilbestrol diphosphate; diethylstilbestryl diphosphate; diethyldihydroxystilbene diphosphate; stilbestrol diphosphate; Stilphostrol (Bayer). C18H22O8P2; mol wt 428.31.
C 50.48%, H 5.18%, O 29.88%, P 14.46%.
Description and references
Synthetic, nonsteroidal estrogen. Prepd by treating stilbestrol with phosphorus oxychloride in pyridine: Miescher, Heer, US 2234311 (1941); Arnold, US 2802854 (1957 to Asta-Werke). Prepn of stable solns at pH 10: Fonner, Collins, US 2828244 (1958 to Miles Labs.). Prepn of sodium salts: Dawson, US 2971975 (1961 to Miles Labs.). Clinical pharmacokinetics: H. Oelschlger et al., Arzneim.-Forsch. 36, 1284 (1986). Review of clinical trials in prostate cancer: J-P. Droz et al, Cancer 71, 1123-1130 (1993).
Properties
Voluminous white cryst powder from dil HCl. Dec 204-206°. Sparingly sol in water.Derivative
Disodium salt.Nomenclature
CAS number: 5965-09-3
C18H20Na2O8P2; mol wt 472.27.
C 45.78%, H 4.27%, Na 9.74%, O 27.10%, P 13.12%.
Properties
Crystals. Softens at 190°, uniform melt at 230°. Sol in water. pH of aq soln 6.5. pH also reported as 4.85 to 5.00.Derivative
Tetrasodium salt.Nomenclature
CAS number: 4719-75-9
Honvan (Baxter); Honvol (Horner); ST-52 (Baxter); Stilbostatin (Taro). C18H18Na4O8P2; mol wt 516.24.
C 41.88%, H 3.51%, Na 17.81%, O 24.79%, P 12.00%.
Therapeutic Category
Antineoplastic (hormonal).
Keywords
Antineoplastic (Hormonal); Estrogens; Estrogen; Nonsteroidal