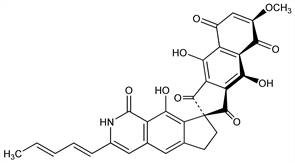
4267. Fredericamycin A
Nomenclature
Description and references
Antitumor antibiotic produced by Streptomyces griseus (FCRC-48); represents new structural class of antibiotics containing a spiro[4,4]nonane ring system. Isoln together with two biologically inactive components, fredericamycins B and C: R. C. Pandey et al., J. Antibiot. 34, 1389 (1981). Antimicrobial and cytotoxic activity in vitro and antitumor activity in vivo: D. J. Warnick-Pickle et al., ibid. 1402. Prepn and biological activity of water soluble salts: R. Misra, ibid. 51, 976 (1988). Structure determn by x-ray crystallography: R. Misra et al., J. Am. Chem. Soc. 104, 4478 (1982). Spectroscopic and mass spectral characterization: eidem, J. Antibiot. 40, 786 (1987). Synthetic studies: A. V. R. Rao et al., Chem. Commun. 1984, 1119; K. A. Parker et al., Tetrahedron Lett. 26, 2181 (1985). Synthesis of (±)-form: T. R. Kelly et al., J. Am. Chem. Soc. 108, 7100 (1986). Biosynthetic study: K. M. Byrne et al., Biochemistry 24, 478 (1985). Mechanism of action study: B. D. Hilton et al., ibid. 25, 5533 (1986).