Nomenclature
CAS number: 98-01-1
2-Furancarboxaldehyde; 2-furaldehyde; pyromucic aldehyde; artificial oil of ants; “furfurol”.
C
5H
4O
2; mol wt 96.08.
C 62.50%, H 4.20%, O 33.30%.
Description and references
Occurs in some essential oils. Prepd industrially
from pentosans which are contained in cereal straws and brans. Laboratory
prepn from corncobs: R. Adams, V. Voorhees, Org. Synth. coll. vol. I, 280 (2nd ed., 1941).
May also be prepd from pyridine. Toxicity study: P. M. Jenner et al., Food Cosmet. Toxicol. 2, 327 (1964).
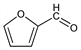
Properties
Colorless oily liq. Peculiar odor, somewhat resembling
the odor of benzaldehyde. Turns yellow to brown on exposure to air
and light and resinifies (the polymerization is greatly accelerated
by hot alkali). d425 1.1563. bp760 161.8°; bp100 103°; bp20 67.8°; bp1.0 18.5° mp 36.5°. Volatile in steam. nD20 1.5261. Absorption spectrum: Purvis, J. Chem. Soc. 97, 1655 (1910).
Sol in 11 parts water; very sol in alcohol, ether. Flash pt, closed cup, 140°F (60°C); open cup, 155°F (68°C). Lower explosive limit: 2.1% by vol in air. Autoignition temp
797°F (392°C). Keep in airtight container
and protect from light. LD50 orally in rats: 127 mg/kg (Jenner).Caution
Potential symptoms of overexposure
are irritation of eyes, skin and upper respiratory system; headache;
dermatitis. See NIOSH Pocket Guide
to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 150.Use
In the manufacture of furfural-phenol plastics such
as Durite; in solvent refining of petroleum oils; in the prepn of
pyromucic acid. As a solvent for nitrated cotton, cellulose acetate,
and gums; in the manuf of varnishes; for accelerating vulcanization;
as insecticide, fungicide, germicide. As a catalyst; as a reagent
in analytical chemistry. In the synthesis of furan derivatives.
