4307. 2-Furoic Acid
Nomenclature
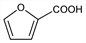
CAS number: 88-14-2
2-Furancarboxylic acid; α-furoic acid; pyromucic acid; Brenzschleimsure (German). C5H4O3; mol wt 112.08.
C 53.58%, H 3.60%, O 42.82%.
Description and references
Prepd from furfural by a Cannizzaro reaction: Wilson, Org. Synth. coll. vol. I (2nd ed., 1941) p 276; cf. US 2041184 (to Quaker Oats), C.A. 30, 4515 (1936); Wojcik, Ind. Eng. Chem. 40, 210 (1948); Harrisson, Moyle, Org. Synth. coll. vol. IV, 493 (1963). Formed in man and several animal species as a metabolite of furfural and related compds.
Properties
Elongated monoclinic prisms from water or by sublimation at 130-140° and 50-60 mm pressure. mp 133-134°. bp760 230-232°; bp20 141-144°. pK (25°) 3.12. Absorption spectrum: Hartley, Dobbie, J. Chem. Soc. 73, 600 (1898). One gram dissolves in 26 ml water at 15°; in 4 ml boiling water. More sol in alcohol; freely sol in ether.Derivative
Ethyl ester.Nomenclature
CAS number: 614-99-3
Ethyl 2-furoate; ethyl pyromucate. C7H8O3; mol wt 140.14.
C 59.99%, H 5.75%, O 34.25%.
Properties
Colorless crystals. d420 1.117. mp 34-36°. bp706 195°. Insol in water; sol in alcohol.Derivative
Methyl ester.Description and references
See Methyl 2-furoate.