4340. Galantamine
Nomenclature
Description and references
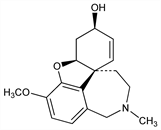
Selective acetylcholinesterase inhibitor. Isoln from Caucasian snowdrops, Galanthus woronowii Vel., Amaryllidaceae: N. F. Proskurnina, A. P. Yakovleva, J. Gen. Chem. USSR 22, 1899 (1952); from Narcissus spp: Boit et al., Ber. 90, 725, 2197 (1957). Structure work: S. Kobayashi et al., Chem. Ind. (London) 1956, 177. Synthesis and stereochemistry: D. H. R. Barton, G. W. Kirby, Proc. Chem. Soc. London 1960, 392; eidem, J. Chem. Soc. 1962, 806. Asymmetric synthesis of isomers from l-tyrosine: K. Shimizu et al., Heterocycles 8, 277 (1977). Biosynthesis studies: D. H. R. Barton et al., J. Chem. Soc. 1963, 4545; W. Dbke, Heterocycles 6, 551 (1977). Toxicology study: S. L. Friess et al., Toxicol. Appl. Pharmacol. 3, 347 (1961). Clinical pharmacokinetics: U. Bickel et al., Clin. Pharmacol. Ther. 50, 420 (1991). Review of pharmacology: A. L. Harvey, Pharmacol. Ther. 68, 113-128 (1995); of clinical experience in Alzheimer's disease: R. Bullock, Expert Rev. Neurother. 4, 153-163 (2004); of synthesis and pharmacology: J. Marco-Contelles et al., Chem. Rev. 106, 116-133 (2006).
Properties
Crystals from benzene, mp 126-127°. [α]D20 118.8° (c = 1.378 in ethanol). Monoacidic base. Fairly sol in hot water; freely sol in alcohol, acetone, chloroform. Less sol in benzene, ether.Derivative
Hydrochloride.Properties
Crystals from water, dec 256-257°. Sparingly sol in cold, more sol in hot water. Very sparingly sol in alcohol, acetone.Derivative
Hydrobromide.Nomenclature
Properties
Crystals from water, dec 246-247°. [α]D20 93.1° (c = 0.1015 in 15 ml H2O). Sparingly sol in water. LD50 i.v. in mice (mg/kg): 5.2 ± 0.2 (Friess).Therapeutic Category
Cholinesterase inhibitor.
Keywords
Cholinesterase Inhibitor