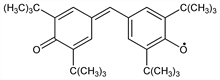
4357. Galvinoxyl
Nomenclature
Description and references
Stable phenoxyl radical. Prepn: G. M. Coppinger, J. Am. Chem. Soc. 79, 501 (1957); M. S. Kharasch, B. S. Joshi, J. Org. Chem. 22, 1435 (1957). Radical scavenging study: P. D. Bartlett, T. Funahashi, J. Am. Chem. Soc. 84, 2596 (1962). Electrical properties: D. D. Eley et al., Trans. Faraday Soc. 62, 3192 (1966). Structure determn: D. E. Williams, Mol. Phys. 16, 145 (1969). Resonance Raman study: G. N. R. Tripathi, Chem. Phys. Lett. 81, 375 (1981). Theoretical study of magnetic interactions: F. Dietz et al., J. Phys. Chem. B 102, 3912 (1998). Ab initio studies of ferromagnetic properties: S. J. Luo, K. L. Yao, J. Magn. Magn. Mater. 257, 11 (2003). Use in detection of phospholipid phase transitions: M. A. Singer et al., Anal. Biochem. 94, 322 (1979). Use in measurement of hydrogen-donating activity and antioxidant activity in phenols: H. Shi et al., Methods Enzymol. 335, 157 (2001).