4369. Garenoxacin
Nomenclature
Description and references
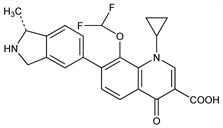
Des-F(6)-quinolone antibacterial; topoisomerase II inhibitor. Prepn: Y. Todo et al., WO 9729102; eidem, US 6025370 (1997, 2000 both to Toyama); and antibacterial activity: K. Hayashi et al., Arzneim.-Forsch. 52, 903 (2002). HPLC determn in serum: D. Xuan et al., J. Chromatogr. B 765, 37 (2001). Mechanism of action study: D. Ince et al., Antimicrob. Agents Chemother. 46, 3370 (2002). Comparative antimicrobial spectra: M. Takahata et al., ibid. 43, 1077 (1999); M. Bassetti et al., ibid. 46, 234 (2002). Clinical pharmacokinetics: D. A. Gajjar et al., ibid. 47, 2256 (2003); and pharmacodynamics: S. Van Wart et al., ibid. 48, 4766 (2004). Toxicology study: A. Nagai et al., J. Toxicol. Sci. 27, 219 (2002). Review of pharmacology and therapeutic potential: R. Frechette, Curr. Opin. Investig. Drugs 2, 1706-1711 (2001).
Properties
Pale yellow plates from ethanol as 0.25 hydrate, mp 226-227°. [α]D27 9.0° (c = 0.10 in N,N-dimethylformamide). Also reported as hydrate, mp 234-235°.Derivative
Methanesulfonate.Nomenclature
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Quinolones and Analogs; Topoisomerase II Inhibitor