4371. Garner's Aldehyde
Nomenclature
CAS number: 127589-93-9
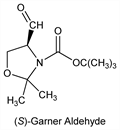
4-Formyl-2,2-dimethyl-3-oxazolidinecarboxylic acid 1,1-dimethylethyl
ester; 1,1-dimethylethyl 4-formyl-2,2-dimethyloxazolidine-3-carboxylate. C11H19NO4; mol wt 229.27.
C 57.63%, H 8.35%, N 6.11%, O 27.91%.
Description and references
Both enantiomers are configurationally stable building blocks for use in asymmetric synthesis. Prepn of R-form: P. Garner, Tetrahedron Lett. 25, 5855 (1984); of S-form: P. Garner, J. M. Park, J. Org. Chem. 52, 2361 (1987). Improved prepn of S-form: A. McKillop et al., Synthesis 1994, 31. Review of chemistry: X. Liang et al., J. Chem. Soc. Perkin Trans. 1 2001, 2136-2157.
Derivative
R-Form.Nomenclature
CAS number: 95715-87-0
(R)-Garner aldehyde. Properties
[α]D27 +103° (c = 1.0 in CHCl3).Derivative
S-Form.Nomenclature
CAS number: 102308-32-7
(S)-Garner aldehyde.