4398. Gentisic Acid
Nomenclature
Description and references
Occurs in gentian: Redgrove, Pharm. J. 122, 324 (1929). Found in urine of dogs after ingestion of salicylates: Neuberg, Berl. Klin. Wochenschr. 48, 799 (1911). Prepd from hydroquinone: Senhofer, Sarlay, Monatsh. Chem. 2, 448 (1881); Juch, ibid. 26, 839 (1905); Brunner, Ann. 351, 321 (1907); Zeltner, Landau, DE 258887; Frdl. 11, 210; Dyson, Manual of Organic Chemistry vol. I (London, 1950) p 614; by oxidation of salicylic acid with potassium persulfate: DE 81297 (to Schering AG); Frdl. 4, 127; from p-hydroxyphenol: Meyer, US 2588336 (1952 to Monsanto); from 5-bromo-2-hydroxybenzoic acid: Lowenthal, Pepper, J. Am. Chem. Soc. 72, 3292 (1950); by Kolbe synthesis: Clemens, US 2816137 (1957 to Eastman Kodak). Metabolic product of Penicillium patulum: Tanenbaum, Bassett, Biochim. Biophys. Acta 28, 21 (1958); of Polyporus tumulosus Cooke: Crowden, Ralph, Aust. J. Chem. 14, 475 (1961). Biosynthesis: Gatenback, Linnroth, Acta Chem. Scand. 16, 2298 (1962).
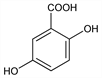
Properties
Needles, monoclinic prisms from water, mp 199-200°. Crystals are dimorphic and undergo phase inversion upon heating. The stable phase begins to sublime at 200° and melts at 205°. pK (25°): 2.93. Sol in water (about 1 part in 200 parts H2O at 5°, much more sol in hot water), alcohol, ether. Practically insol in carbon disulfide, chloroform, benzene.Derivative
Sodium salt.Nomenclature
Properties
Crystals with 5H2O from water. Rapidly loses 3H2O on exposure to air, but holds H2O tenaciously even at 100°. Sol in water.Therapeutic Category
Analgesic; anti-inflammatory.
Keywords
Anti-inflammatory (Nonsteroidal); Salicylic Acid Derivatives; Analgesic; Non-opioids; Salicylic Acid Derivatives