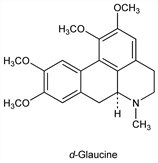
4436. Glaucine
Nomenclature
CAS number: 475-81-0
(6aS)-5,6,6a,7-Tetrahydro-1,2,9,10-tetramethoxy-6-methyl-4H-dibenzo[de,g]quinoline; 1,2,9,10-tetramethoxyaporphine; boldine dimethyl ether; Bromcholitin; Glauvent. C21H25NO4; mol wt 355.43.
C 70.96%, H 7.09%, N 3.94%, O 18.01%.
Description and references
d-Form prevalent in nature. Found in Glaucium flavum Crantz (G. luteum Scop.), Papaveraceae and in Dicentra and Corydalis species, Fumariaceae. Isoln: Fischer, Arch. Pharm. 239, 426 (1901). Structure and prepn: Gadamer, ibid. 249, 680 (1911); Spth, Tharrer, Ber. 66, 904 (1933). Configuration: Faltis, Adler, Arch. Pharm. 284, 281 (1951). Synthesis of dl-form: Chan, Maitland, J. Chem. Soc. C 1966, 753; Jackson, Martin, ibid. 2061; Cava et al., J. Org. Chem. 35, 175 (1970). Pharmacology and toxicity of dl-glaucine phosphate: Y. Kasé et al., Arzneim.-Forsch. 33, 936 (1983). Sites of antitussive action: eidem, ibid. 947.
Properties
Orthorhombic plates, prisms from ethyl acetate or ether, mp 120°. [α]D20 +115° (c = 3 in alc). Sol in acetone, alcohol, chloroform, ethyl acetate. Moderately sol in ether, petr ether. Practically insol in water and benzene.Derivative
Hydrochloride trihydrate.C21H25NO4.HCl.3H2O; mol wt 445.93.
C 56.56%, H 7.23%, N 3.14%, O 25.12%, Cl 7.95%.
Properties
Needles. mp 232° (anhydrous). Sol in water, alcohol, chloroform.Derivative
Hydrobromide.C21H25NO4.HBr; mol wt 436.34.
C 57.80%, H 6.01%, N 3.21%, O 14.67%, Br 18.31%.
Properties
Crystals, mp 235°. Less sol than the hydrochloride.Derivative
dl-Form phosphate.Nomenclature
DL-832.(C21H25NO4)2.3H3PO4; mol wt 1004.84.
C 50.20%, H 5.92%, N 2.79%, O 31.84%, P 9.25%.