4447. Glucagon-Like Peptides
Description and references
Bioactive peptides (GLP-1 and GLP-2) encoded within the preproglucagon gene; formed by tissue-specific, post-translational processing of proglucagon by intestinal L-cells. Key regulators of energy homeostasis and metabolism. Released into the circulation in response to a meal; rapidly inactivated by dipeptidylpeptidase-IV (DPP-IV). GLP-1 is a potent, glucose-dependent stimulator of insulin secretion (incretin). GLP-2 has growth-promoting and cytoprotective effects on intestinal epithelium. Identification in hamster preproglucagon: G. I. Bell et al., Nature 302, 716 (1983); in human: idem et al., ibid. 304, 368 (1983). Review of structures, tissue distribution, and physiological activites: T. J. Kieffer, J. F. Habener, Endocr. Rev. 20, 876-913 (1999). Review of biosynthesis and regulation of secretion: P. L. Brubaker, Y. Anini, Can. J. Physiol. Pharmacol. 81, 1005-1012 (2003). Review of bioactivities: L. L. Baggio, D. J. Drucker, Best Pract. Res. Clin. Endocrinol. Metab. 18, 531-554 (2004); and therapeutic potential: D. J. Drucker, Nat. Clin. Pract. Endocrinol. Metab. 1, 22-31 (2005).
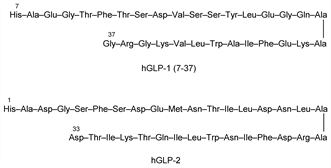
Derivative
GLP-1.Nomenclature
Description and references
Originally identified as a 37 amino acid peptide; bioactive forms are GLP-1 (7-37) and GLP-1 (7-36) amide. Stimulates insulin secretion, suppresses glucagon secretion, and delays gastric emptying. Review of physiology and therapeutic potential in diabetes: M. A. Nauck, Acta Diabetol. 35, 117-129 (1998); M. M. J. Combettes, Curr. Opin. Pharmacol. 6, 598-605 (2006).
Derivative
GLP-2.Nomenclature
Description and references
Intestinotrophic peptide containing 33 amino acid residues. Stimulates crypt-cell proliferation and inhibits apoptosis in the gastrointestinal mucosa. Identification of bioactivity: D. J. Drucker et al., Proc. Natl. Acad. Sci. USA 93, 7911 (1996). Comprehensive review: J. L. Estall, D. J. Drucker, Annu. Rev. Nutr. 26, 391-411 (2006).