4463. Glucosulfone Sodium
Nomenclature
CAS number: 554-18-7
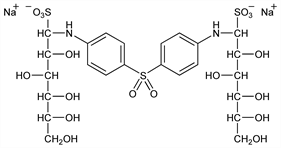
1,1′-[Sulfonylbis(4,1-phenyleneimino)]bis[1-deoxy-1-sulfo-d-glucitol] disodium salt; p,p′-sulfonyldianiline N,N′-diglucoside
disodium disulfonate; p,p′-sulfonyldianiline-N,N′-di-d-glucose sodium bisulfite compd; p,p′-diaminodiphenylsulfone-N,N′-di(dextrose
sodium sulfonate); disodium p,p′-diaminodiphenylsulfone-N,N′-diglucose sulfonate; 501-P; Protomin; Promin (Parke-Davis); Promanide. C24H34N2Na2O18S3; mol wt 780.70.
C 36.92%, H 4.39%, N 3.59%, Na 5.89%, O 36.89%, S 12.32%.
Description and references
Prepd by refluxing a mixture of 4,4′-diaminodiphenylsulfone, glucose, sodium bisulfite, and 80% ethanol: CH 234108 (1944 to B. Siegfried), C.A. 43, 4297a (1949); B. C. Jain et al., Sci. Cult. 11, 568 (1946). Structure activity study: W. Logemann, G. P. Miori, Arzneim.-Forsch. 5, 213 (1955). Clinical evaluation in ocular leprosy: A. Bouzas, Arch. Ophthalmol. 31, 629 (1971).
Properties
White, amorphous powder. Soluble in water; slightly sol in ethanol. Insol in ether, benzene, methanol, ethyl acetate, pyridine. Aq solns may be sterilized by autoclaving. LD50 in rats (g/kg): 3-4 orally, 3-3.5 i.v. (Logemann, Miori).Therapeutic Category
Antibacterial (leprostatic).
Keywords
Antibacterial (Synthetic); Sulfones; Antibacterial (Leprostatic)