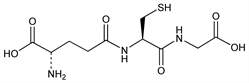
4475. Glutathione
Nomenclature
Description and references
The major low mol wt thiol compound of the living plant or animal cell. Isoln from yeast: Hopkins, J. Biol. Chem. 84, 269 (1929). Synthesis: du Vigneaud, Miller, Biochem. Prep. 2, 87 (1952); Goldschmidt et al., Ber. 97, 2434 (1964); Y. Ozawa et al., Bull. Chem. Soc. Jpn. 53, 2592 (1980). Review of early syntheses: Jeschkeit et al., Pharmazie 18, 658 (1963). Review of metabolism: A. Meister, M. E. Anderson, Annu. Rev. Biochem. 52, 711-760 (1983); of metabolic role in antineoplastic chemotherapy: B. A. Arrick, C. F. Nathan, Cancer Res. 44, 4224-4232 (1984). Monographs: S. Colowick et al., Glutathione (Academic Press, New York, 1954); Glutathione, E. M. Crook, Ed., Biochem. Soc. Symposium No. 17, London, 1958 (Cambridge University Press, 1959); Glutathione, L. Flohe et al., Eds. (Academic Press, New York, 1974); Glutathione: Metabolism & Function I. M. Arias, W. B. Jackoby, Eds. (Raven, New York, 1976).