4554. Guaiazulene
Nomenclature
CAS number: 489-84-9
1,4-Dimethyl-7-(1-methylethyl)azulene; 7-isopropyl-1,4-dimethylazulene; S-guaiazulene; AZ 8 (Millet Roux); AZ 8 Beris (Weimer); Eucazulen; Kessazulen; Vaumigan. C15H18; mol wt 198.30.
C 90.85%, H 9.15%.
Description and references
Isoln from chamomile oil: Sorm et al., Collect. Czech. Chem. Commun. 16, 626 (1951); from guaiac wood oil: Joos, CH 314487 (1956), C.A. 52, 443b (1958). Total synthesis: Plattner et al., Helv. Chim. Acta 32, 2452 (1949); Sorm et al., Collect. Czech. Chem. Commun. 16, 168 (1951); Jacob et al., Tetrahedron 20, 2821 (1964); J. Mukherjee et al., J. Am. Chem. Soc. 101, 251 (1979). Pharmacokinetics of guaiazulene soluble in animals: H. Mukai et al., J. Pharmacobio-Dyn. 8, 329, 337 (1985). Effect on gastric and duodenal ulcers in rats: S. Okabe et al., Nippon Yakurigaku Zasshi 88, 467 (1986), C.A. 106, 43769 (1987).
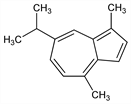
Properties
Blue oil. bp10 165-170°.Derivative
3-Sulfonate sodium salt.Nomenclature
CAS number: 6223-35-4
5-Isopropyl-3,8-dimethyl-1-azulenesulfonic acid sodium salt; sodium gualenate; guaiazulene soluble; Azulon (Rorer). C15H17NaO3S; mol wt 300.35.
C 59.98%, H 5.71%, Na 7.65%, O 15.98%, S 10.68%.
Derivative
Trinitrobenzene deriv.Nomenclature
CAS number: 4968-29-0
C15H18.C6H3N3O6; mol wt 411.41.
C 61.31%, H 5.14%, N 10.21%, O 23.33%.
Properties
Violet to black needles from ethanol, mp 151°.Therapeutic Category
Anti-inflammatory; antiulcerative.
Keywords
Anti-inflammatory (Nonsteroidal); Antiulcerative; Cytoprotectant (Gastric)