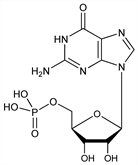
4570. 5′-Guanylic Acid
Nomenclature
Description and references
Nucleotide widely distributed in nature; found in hydrolyzates of RNA. Isolated together with inosinic acid from sardines or yeast extract: Kuninaka et al., New Food Ind. 3, no. 1, 21 (1961). Also by direct biosynthesis using microorganisms or enzymes: Abrams, Bentley, Arch. Biochem. Biophys. 79, 91 (1959); Magasanik, Karibian, J. Biol. Chem. 235, 2672 (1960); Okumura et al., US 3249511 (1966). Chemical synthesis: Michelson, Todd, J. Chem. Soc. 1949, 2483; Chambers et al., J. Am. Chem. Soc. 79, 3747 (1957); Gilham, Tener, Chem. Ind. (London) 1959, 542; Tener, J. Am. Chem. Soc. 83, 159 (1961); Koransky et al., Z. Naturforsch. 17B, 291 (1962). Prepn of Na salt: Ishibashi, Ito, US 3190877 (1965 to Takeda). Monograph on synthesis of nucleotides: G. R. Pettit, Synthetic Nucleotides vol. 1 (Van Nostrand Reinhold, New York, 1972) 252 pp. Reviews: See Guanidine.