4611. Haplophytine
Nomenclature
CAS number: 16625-20-0
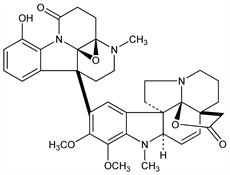
[15(3aS,7R)]-3,4-Didehydro-19-hydroxy-16,17-dimethoxy-1-methyl-15-(2,3,5,6-tetrahydro-11-hydroxy-4-methyl-1,13-dioxo-1H-3a,7-methanopyrrolo[1,2-a][1,3]benzodiazocin-7(4H)-yl)aspidospermidin-21-oic acid γ-lactone. C37H40N4O7; mol wt 652.74.
C 68.08%, H 6.18%, N 8.58%, O 17.16%.
Description and references
Insecticidal alkaloid from the Mexican plant Haplophyton cimicidum A. DC., Apocynaceae: Rogers et al., J. Am. Chem. Soc. 74, 1987 (1952); 76, 2819 (1954). Partial structure: Snyder et al., ibid. 80, 3708 (1958). Structure: Rae et al., ibid. 89, 3061 (1967). Crystal structure and absolute config.: Zacharias, Acta Crystallogr. 26B, 1455 (1970). Synthetic approaches: P. Yates, D. A. Schwartz, Can. J. Chem. 61, 509 (1983). Review: Yates et al., J. Am. Chem. Soc. 95, 7842 (1973).
Properties
Crystals from ethanol + chloroform, mp 290-293° (rapid heating, starting at 250°). Also reported as mp 300-302° (Yates et al., loc. cit.). [α]D25 +109.0° (chloroform). uv max (ethanol): 220, 265, 305 nm (ε 48500, 14300, 4500). Very sol in chloroform, benzene, dioxane, ethyl acetate. Moderately sol in acetone, methanol, somewhat less in ethanol. Practically insol in water, ether, petr ether. Readily sol in dil acids or alkalies.Derivative
Dihydrochloride.C27H31N3O5.2HCl; mol wt 550.47.
C 58.91%, H 6.04%, N 7.63%, O 14.53%, Cl 12.88%.
Properties
Dec 208-218° (darkens at 200°).Derivative
O-Methylhaplophytine.C28H33N3O5; mol wt 491.58.
C 68.41%, H 6.77%, N 8.55%, O 16.27%.