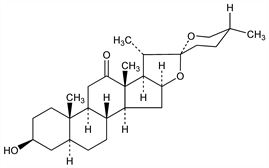
4622. Hecogenin
Nomenclature
CAS number: 467-55-0
(3β,5α,25R)-3-Hydroxyspirostan-12-one. C27H42O4; mol wt 430.62.
C 75.31%, H 9.83%, O 14.86%.
Description and references
A steroidal sapogenin which has been isolated from plants, particularly from numerous Agave species. Isoln: Marker et al., J. Am. Chem. Soc. 69, 2167 (1947); Rubin, US 3303186 (1967). Synthesis and configuration: Mazur et al., J. Am. Chem. Soc. 82, 5889 (1960). Separation of an optically inactive product from sapogenin mixtures: Cardenas, US 3013010 (1961 to Searle). Review: L. F. Fieser, M. Fieser, Steroids (Reinhold, New York, 1959) pp 667-671 sqq.
Properties
Crystals from acetone, mp 264-266°. [α]D +8° (CHCl3) (Mazur et al.); mp 245°, 253°, 268° (Marker et al.); mp 240-245°, 245-250°. [α]D ±0° (CHCl3) (Cardenas).Derivative
Pseudohecogenin.Nomenclature
CAS number: 11005-20-2
(3β,5α,25R)-3,26-Dihydroxyfurost-20(22)-en-12-one. C27H42O4; mol wt 430.62.
C 75.31%, H 9.83%, O 14.86%.
Description and references
Prepn from hecogenin: Cameron et al., J. Chem. Soc. 1955, 2807; Wall, Serota, US 2870143 (1959 to USDA).