4633. Helvolic Acid
Nomenclature
CAS number: 29400-42-8
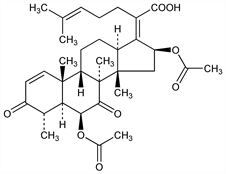
(4α,6β,8α,9β,13α,14β,16β,17Z)-6,16-Bis(acetyloxy)-3,7-dioxo-29-nordammara-1,17(20),24-trien-21-oic
acid; (Z)-6β,16β-dihydroxy-3,7-dioxo-29-nor-8α,9β,13α,14β-dammara-1,17(20),24-trien-21-oic
acid diacetate; fumigacin. C33H44O8; mol wt 568.70.
C 69.69%, H 7.80%, O 22.51%.
Description and references
Antibiotic substance of the fusidane class. Produced by Aspergillus fumigatus: Waksman et al., J. Bacteriol. 45, 233 (1943); by A. fumigatus mut. helvola: Chain et al., Br. J. Exp. Pathol. 24, 108 (1943). Structural studies: Okuda et al., Chem. Pharm. Bull. 12, 121 (1964); Oxley, Chem. Commun. 1966, 729; Okuda et al., Tetrahedron Lett. 1967, 2295. Revised structure and stereochemistry: Iwaki et al., Chem. Commun. 1970, 1119. Chemical and biological data: Reshetova, Antibiotiki 14, 554 (1969). Review of literature until 1960: Wilson, “Miscellaneous Aspergillus Toxins” in Microbial Toxins vol. VI, A. Ciegler et al., Eds. (Academic Press, New York, 1971) p 265.
Properties
Needles from methanol, mp 215°. [α]D25 121° (chloroform). uv max (ethanol): 231 nm (log ε 4.24). Very slightly sol in water; slightly sol in petr ether, methanol, ethanol. More sol in hot methanol, ethanol; sol in chloroform, acetone, ethyl acetate, benzene, pyridine, glacial acetic acid, dioxane, ether.Derivative
Methyl ester.Nomenclature
Methyl helvolate.C34H46O8; mol wt 582.72.
C 70.08%, H 7.96%, O 21.97%.