4644. Hemin
Nomenclature
CAS number: 16009-13-5
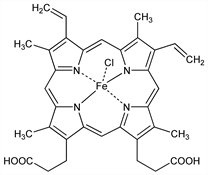
(SP-5-13)-Chloro[7,12-diethenyl-3,8,13,17-tetramethyl-21H,23H-porphine-2,18-dipropanoato(4)-N21,N22,N23,N24]ferrate(2) dihydrogen; chloro[dihydrogen 3,7,12,17-tetramethyl-8,13-divinyl-2,18-porphinedipropionato(2)]iron; chlorohemin; 1,3,5,8-tetramethyl-2,4-divinylporphine-6,7-dipropionic acid
ferrichloride; Teichmann's crystals; ferriheme chloride; ferriprotoporphyrin chloride; ferriporphyrin chloride. C34H32ClFeN4O4; mol wt 651.94.
C 62.64%, H 4.95%, Cl 5.44%, Fe 8.57%, N 8.59%, O 9.82%.
Description and references
Prepd from hemoglobin soln by heating with acetic acid and sodium chloride. Practical procedure for its prepn from ox blood: Schalfejeff, J. Russ. Phys. Chem. Soc. 1885, 30; Ber. 18, 232 (1885); Gattermann-Wieland, Praxis des Organischen Chemikers 23rd ed., p 407; Fischer-Orth, Die Chemie des Pyrrols II, 1, 377 (Leipzig, 1937); H. Fischer, Org. Synth. coll. vol. III, 442 (1955); Labbe, Nishida, Biochim. Biophys. Acta 26, 437 (1957). Biosynthesis: Karlzeile, Angew. Chem. 66, 729 (1954). Review: Stoll, Experientia 4, 6 (1948); H. H. Inhoffen, Naturwissenschaften 55, 457 (1968); W. S. Caughey in Inorganic Biochemistry vol. 2, G. L. Eichhorn, Ed. (Elsevier, New York, 1973) pp 797-831.
Properties
Long, thin blades from glacial acetic acid or from chloroform-pyridine-acetic acid, appearing brown in transmitted light and steel-blue in reflected light. Sinters at 240° but is not melted at 300°. Absorption spectrum: Fischer-Orth, loc. cit. Freely sol in dil ammonia water, also in solns of sodium hydroxide with hematin formation, i.e., the chlorine is displaced by an OH group. Practically insol in carbonate solns, dil acid solns. Sol in strong organic bases such as trimethylamine, p-toluidine, dimethylaniline. Sol in concd H2SO4 with loss of Fe. Sparingly sol in 70-80% alc. Practically insol but stable in water.Derivative
Dimethyl ester.C36H36ClFeN4O4; mol wt 679.99.
C 63.59%, H 5.34%, Cl 5.21%, Fe 8.21%, N 8.24%, O 9.41%.
Properties
Needles from benzene, not melted at 300°. Freely sol in acetic acid, benzene, chloroform, acetone.Derivative
Diethyl ester.C38H40ClFeN4O4; mol wt 708.05.
C 64.46%, H 5.69%, Cl 5.01%, Fe 7.89%, N 7.91%, O 9.04%.