4685. Hexafluoroacetone
Nomenclature
CAS number: 684-16-2
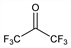
1,1,1,3,3,3-Hexafluoro-2-propanone. C3F6O; mol wt 166.02.
C 21.70%, F 68.66%, O 9.64%.
Description and references
Synthon in organic and inorganic reactions with an exceptionally electron deficient carbonyl group. Prepn: N. Fukuhara, L. A. Bigelow, J. Am. Chem. Soc. 63, 788 (1941). Comprehensive review: C. G. Krespan, W. J. Middleton in Fluorine Chem. Rev. vol. 1, P. Tarrant, Ed. (Marcel Dekker, New York, 1967) pp 145-196. Review of reactivity: H. W. Roesky, J. Fluorine Chem. 30, 123-139 (1985); of use in inorganic chemistry: M. Witt et al., Adv. Inorg. Chem. Radiochem. 30, 223-312 (1986); in polymer chemistry: P. E. Cassidy et al., J. Macromol. Sci. Rev. Macromol. Chem. Phys. C29, 365-429 (1989); in peptide chemistry: K. Burger et al., Amino Acids 8, 195-199 (1995); of toxicology: G. L. Kennedy, Jr., Crit. Rev. Toxicol. 21, 149-170 (1990).