4715. HHCB
Nomenclature
CAS number: 1222-05-5
1,3,4,5,6,7,8-Hexahydro-4,6,6,7,8,8-hexamethylcyclopenta[g]-2-benzopyran; 6-oxa-1,1,2,3,3,8-hexamethyl-2,3,5,6,7,8-hexahydro-1H-benz[f]-indene; Galaxolide (Int. Flavors & Fragrances). C18H26O; mol wt 258.40.
C 83.67%, H 10.14%, O 6.19%.
Description and references
Isochroman musk odorant; scent is primarily due to the ()-(4S,7R) and ()-(4S,7S) isomers. Prepn: L. G. Heeringa, M. G. J. Beets, GB 991146; eidem, US 3360530 (1965, 1967 both to International Flavors & Fragrances). Prepn and olfactory characterization of enantiomers: G. Fráter et al., Helv. Chim. Acta 82, 1656 (1999). Synthesis of olfactorally active stereoisomers: A. Ciappa et al., Tetrahedron: Asymmetry 13, 2193 (2002). HPLC determn: W. Schüssler, L. Nitschke, Fresenius J. Anal. Chem. 361, 220 (1998). Subchronic toxicity study: A. M. Api, R. A. Ford, Toxicol. Lett. 111, 143 (1999).
Properties
Colorless crystals from methanol, mp 57-58°. bp0.8 129°. Strong musk odor. nD20 1.5342. d420 1.0054. Crystal density: 1.087 g/cm3.Derivative
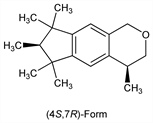
()-(4S,7R)-Form.Nomenclature
CAS number: 252332-95-9
Properties
Colorless crystals from methanol, mp 78-78.5°. [α]D24 23.5°; [α]54624 27.3° (c = 5.0 in ethanol). Powerful typical musk odor.Derivative
()-(4S,7S)-Form.Nomenclature
CAS number: 172339-62-7
Properties
Colorless crystals from hexane, mp 57-58°. [α]D23 25.1°; [α]54623 29.2° (c = 5.0 in ethanol). Musky odor with dry aspects; slightly less powerful than ()-(4S,7R)-form.Derivative
(+)-(4R,7S)-Form.Nomenclature
CAS number: 172339-63-8
Properties
Colorless crystals from methanol, mp 77-78°. [α]D24 +24.1°; [α]54624 +28.0° (c = 5.2 in ethanol). Weak, musky, uncharacteristic odor.Derivative
(+)-(4R,7R)-Form.Nomenclature
CAS number: 252332-96-0