4724. HL-7
Nomenclature
Description and references
Bisquaternary Hagedorn oxime; acetylcholinesterase reactivator. In vitro reactivation of tabun and soman acetylcholinesterase inhibition: L. P. A. de Jong et al., Biochem. Pharmacol. 38, 633 (1989). Stability and decomposition: P. Eyer et al., Arch. Toxicol. 63, 59 (1989). Synthesis, pharmacology and toxicity: eidem, ibid. 66, 603 (1992). Pharmacokinetics: U. Sphrer et al., ibid. 68, 480 (1994). Efficacy in nerve agent poisoning: B. P. C. Melchers et al., Pharmacol. Biochem. Behav. 49, 781 (1994). MS determn: P. A. D'Agostino et al., Rapid Commun. Mass Spectrom. 10, 805 (1996). Comparative reactivation studies in human acetylcholinesterase inhibition: F. Worek et al., Biochem. Pharmacol. 68, 2237 (2004).
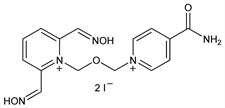
Properties
Crystals from hot water, mp 160° (dec). Soly in water: 20 mg/ml. Aq solns are unstable and liberate iodine even when protected from light. uv max (pH 2): 298, 252, 224 nm ( log ε 4.20, 4.20, 4.15); (pH 11) 368 nm (log ε 4.35). pKa1 7.04, pKa2 8.52. LD50 i.m. in female mice: 744 μmol/kg (Eyer, 1992).Derivative
Dimethanesulfonate.Nomenclature
Properties
Crystals from warm methanol, 173-174 (dec). pH (1% aq soln) ≈4. Soly in water: 0.68 g/ml. LD50 i.m. in female mice: 799 μmol/kg (Eyer, 1992).Therapeutic Category
Antidote (organophosphate poisoning).
Keywords
Antidote (Organophosphate Poisoning); Cholinesterase Reactivator