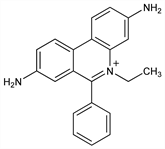
4731. Homidium
Nomenclature
Description and references
Prepn: T. I. Watkins, J. Chem. Soc. 1952, 3059; Short et al., US 2662082 (1953 to Boots Pure Drug). uv spectrum: B. Hudson, R. Jacobs, Biopolymers 14, 1309 (1975). Inhibition of DNA synthesis: B. A. Newton, J. Gen. Microbiol. 17, 718 (1957); of DNA polymerase: W. H. Elliott, Biochem. J. 86, 562 (1963). Intercalcation of double-stranded DNA: M. J. Waring, J. Mol. Biol. 13, 269 (1965); J.-B. LePecq, C. Paoletti, ibid. 27, 87 (1967). Mutagenicity studies: J. T. MacGregor, I. J. Johnson, Mutat. Res. 48, 103 (1977); G. S. Probst et al., Environ. Mutagen. 3, 11 (1981). Review of interactions with nucleic acids: J.-B. LePecq in Methods of Biochemical Analysis vol. 20, D. Glick, Ed. (Wiley-Interscience, New York, 1971) pp 41-86; M. Waring in Antibiotics vol. 3, J. W. Corcoran, F. E. Hahn, Eds. (Springer-Verlag, New York, 1975) pp 141-165.