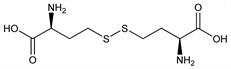
4735. l-Homocystine
Nomenclature
Description and references
Naturally occurring, dimeric form of homocysteine, q.v. Appearance in urine is indicative of the metabolic disorder, cystathionine β-synthase deficiency. Prepn of dl-form from methionine: L. W. Butz, V. du Vigneaud, J. Biol. Chem. 99, 135 (1932). Synthesis by the “malonate” method: W. I. Patterson, V. du Vigneaud, ibid. 111, 393 (1935); from 3,6-bis(β-chloroethyl)-2,5-dioxopiperazine: H. R. Snyder, G. W. Cannon, J. Am. Chem. Soc. 66, 511 (1944) DOI. Prepn of d- and l-forms: V. du Vigneaud, W. I. Patterson, J. Biol. Chem. 109, 97 (1935); H. Miyazaki et al., Bull. Chem. Soc. Jpn. 66, 536 (1993) DOI. Asymmetric synthesis: M. Adamczyk et al., Tetrahedron: Asymmetry 10, 4151 (1999) DOI. Isotachophoretic determn in urine: N. Mizobuchi et al., J. Chromatogr. B 382, 321 (1986) DOI PubMed. LC-MS/MS determn in plasma: M. Tomaiuolo et al., ibid. 842, 64 (2006) DOI PubMed.