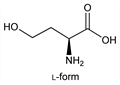
4739. Homoserine
Nomenclature
CAS number: 672-15-1
2-Amino-4-hydroxybutanoic acid; 2-amino-4-hydroxybutyric acid; α-amino-γ-hydroxy-n-butyric acid. C4H9NO3; mol wt 119.12.
C 40.33%, H 7.62%, N 11.76%, O 40.29%.
Description and references
Principal free amino acid occurring in pea plants: A. I. Virtanen, Acta Chem. Scand. 7, 1423 (1953); J.A. Bakhuis, Nature 180, 713 (1957). Prepn: Fischer, Blumenthal, Ber. 40, 106 (1907); Armstrong J. Am. Chem. Soc. 70, 1756 (1948); Birnbaum, Greenstein, Arch. Biochem. Biophys. 42, 212 (1953); M. Frankel, Y. Knobler, J. Am. Chem. Soc. 80, 3147 (1958). Review of homoserine production by fermentation: T. Nara in Microbial Prod. Amino Acids, K. Yamada, Ed. (Wiley, New York, 1972) pp 417-434.