4746. Hordenine
Nomenclature
CAS number: 539-15-1
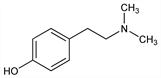
4-[2-(Dimethylamino)ethyl]phenol; N,N-dimethyltyramine; p-hydroxy-N,N-dimethylphenethylamine; anhaline; eremursine; peyocactine. C10H15NO; mol wt 165.23.
C 72.69%, H 9.15%, N 8.48%, O 9.68%.
Description and references
Isoln from barley germs: Leger, Compt. Rend. 142, 108 (1906); 143, 234, 916 (1906); 144, 488 (1907); Erspamer, Falconieri, Naturwissenschaften 39, 431 (1952). Structure: Leger, Bull. Soc. Chim. Fr. [3] 35, 868 (1906); [4] 1, 148 (1907); Gaebel, Arch. Pharm. 244, 441 (1906). Synthesis from phenethyl alcohol: Barger, J. Chem. Soc. 95, 2193 (1909); from tyrosine: Raoul, Compt. Rend. 204, 74 (1937); from p-(β-hydroxyethyl)anisole: Cheng et al., J. Am. Chem. Soc. 73, 4081 (1951).
Properties
Orthorhombic prisms from alcohol or from benzene + petr ether, needles from water, mp 117-118°. bp11 173°. Sublimes 140-150°. Very sol in alcohol, chloroform, ether. 7 grams dissolve in 1000 ml water. Sparingly sol in benzene, toluene, xylene. Practically insol in petr ether.Derivative
Hydrochloride.Nomenclature
CAS number: 6027-23-2
C10H15NO.HCl; mol wt 201.69.
C 59.55%, H 8.00%, N 6.94%, O 7.93%, Cl 17.58%.